Preimplantation Genetic Diagnosis: PGD
What is preimplantation genetic diagnosis (PGD)?
Preimplantation genetic diagnosis is genetic screening technique which allows an expert to examine the genetic code of a pre-embryo before it is transferred to the patient’s uterus. The earliest form of PGD was developed in the late 1980s, and since then the techniques have been greatly refined and enhanced. It is now possible to look at all 23 pairs of chromosomes with very little risk to the embryo.
During IVF, embryos are created in the lab and incubated for a few days. When the embryos are sufficiently mature, one or more is selected and implanted in the patient’s uterus during the embryo transfer procedure. In a routine IVF cycle, the embryos are selected using visual criteria: the embryo or embryos that look strongest and healthiest are the ones chosen for transfer. In some cases, however, a visual inspection is not enough. Some serious genetic abnormalities are invisible to the eye. This is where PGD can be used.
What do infertility clinics test for in genetic testing?
A healthy human genetic code consists of 23 pairs of chromosomes, and each of the chromosomes may contain hundreds or thousands of genes. Half of this genetic material is inherited from the mother and half from the father, and the combination creates the “blueprint” for their offspring. Unfortunately, the process does not always work seamlessly. Problems with the chromosomes or specific gene can cause a pregnancy to miscarry or result in serious genetic conditions and diseases. PGD can be used to identify these issues before a pregnancy occurs. PGD can:
- Detect abnormalities in chromosomes
- Aneuploidy is an imbalanced number of chromosomes. Most embryos with aneuploidy do not survive to term. It is a leading cause of early embryonic demise and spontaneous miscarriage. Missing, extra, or unpaired chromosomes can cause severe genetic disorders and birth defects. The most common survivable aneuploidy is Down syndrome. Also known as Trisomy 21, it occurs when an embryo has three copies of chromosome 21 instead of the usual two.
- Numerical mistakes in the sex chromosomes. The last pair of chromosomes determines sex. A female embryo has two X chromosomes, while a male embryo has an X and a Y chromosome. A missing or extra chromosome here can create a number of syndromes. Symptoms vary according to the affected individual. The most common are:
- Turner’s syndrome (XO) where a female is missing all or part of an X chromosome.
- Klinefelter’s syndrome (XXY) where a male has two or more X chromosomes.
- Translocation is when chromosomes break apart and attach to other chromosomes, which can result in missing or extra genes. This can cause miscarriages in many cases. Some forms of leukemia and cancer are caused by translocations.
- Detect hereditary conditions: Several devastating conditions are caused by specific genes. PGD can determine whether an embryo has inherited these genes. The most common conditions tested for include:
- Cystic fibrosis
- Tay-Sachs disease
- Duchenne muscular dystrophy
- Sickle-cell anemia
- X-chromosome-linked disorders
PGD procedure: how is PGD performed?
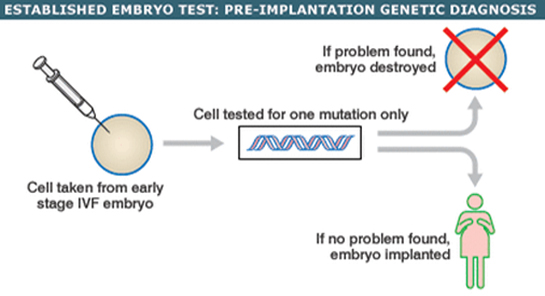
In order to perform PGD, pre-embryos must be available for testing. The patient goes through a regular IVF cycle. Fertility hormones stimulate the ovaries into producing more eggs. When the eggs are mature, they are retrieved from the ovaries in a procedure performed in the clinic under light sedation. Then, in the lab, eggs and sperm are combined and fertilization occurs. The resulting pre-embryo can then be tested.
During the PGD procedure, an embryologist carefully removes a few cells from the embryo. These cells, called blastomeres, are then evaluated for abnormalities. The evaluation normally takes 7-10 days. Once all of the pre-embryos are tested, only healthy, normal embryos are selected for transfer to establish pregnancy in a later cycle.
Who can benefit from PGD?
PGD is not necessary in all cases. Your physician will advise you whether or not it is indicated for your specific situation. PGD is often recommended for:
- Women age 35 and older because there is a concern about the genetic quality of their eggs
- Women experiencing recurrent, unexplained pregnancy loss
- Women with more than one failed fertility treatment
- Carriers of sex-linked disorders – they can choose gender determination to prevent sex-linked disease
- Carriers of single gene disorders
- Couples with family history of inherited disease
- Those with chromosomal disorders
What to consider before choosing PGD
There are many potential benefits of PGD, but like every decision you make about your fertility treatment, it requires careful consideration. For couples confronted with a family history of serious or even life threatening genetic diseases, the decision may be easy. For a patient with less pressing concerns, a review of the other benefits and the risks of PGD may be helpful.
- Benefits of PGD
- Reduces the requirement for amniocentesis later in the pregnancy. Amniocentesis is prenatal testing done when the fetus is aged 10-16 weeks, where a needle is used to obtain a sample of amniotic fluid. As it can be uncomfortable and carries some risk, avoiding this test may be a valuable benefit for some patients.
- Enables the implantation of only selected quality pre-embryo(s). This increases the chance of pregnancy and decreases chance of miscarriage.
- Decreases chance of multiple births. Because they are screened for quality, fewer embryos can be transferred. This helps you avoid the risks and complication of a multiple pregnancy and birth.
- If you are already going through IVF, you have the option to choose the gender.
- Risks of PGD
- Some genetic disorders may not show up until a later stage in life. PGD is highly effective, but in rare cases a defective embryo may not show the disease or defect until mature.
- Moral/Ethical considerations
- Choosing what to do with remaining embryos. Deciding what to do with healthy or unhealthy that aren’t implanted can be an intense decision for some patients. If you hope to try again at a later date, the healthy embryos that remain can be frozen for storage. With unhealthy embryos, you may have the choice to donate them to research or simply decide that they should be discarded.
- Worries about a slippery slope to ‘designer babies.’ Casual discussions of PGD in the media often bring up the idea that genetic testing will lead to embryos that are selected for specific gene traits such as intelligence or physical fitness, leading to accusations of eugenics. In reality, there is no current scientific basis for this assumption. In most cases we don’t know exactly which genes control such traits, or how to turn them “on.” Even if it were possible, the question of nature versus nurture remains: genetics are not the whole story of a human being, and our environment plays a huge role in how each person develops.
If you have reason to believe that you will want to use PGD as a part of your IVF process, it is important to select a clinic with a strong reputation for the procedure. While PGD can be performed safely without harm to developing embryos, any handling or manipulation of an embryo carries very low risk of injury. PGD is a rapidly developing technology, and the benefits of a cutting-edge fertility laboratory and extensively experienced staff are crucial to the success of PGD and the safety of the embryo. For many couples, PGD offers them a chance to have healthy biological children that they could never have had otherwise. In the hands of an expert clinic, the benefits of PGD greatly outweigh the risk.
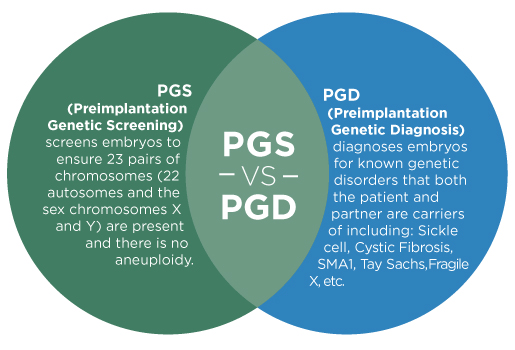
Pre-implantation genetic screening (PGS)
PGS is a technique for testing whether embryos have any problems with their chromosomes, which is thought to be the most common reason IVF treatment fails
What is PGS?
PGS (also known as aneuploidy screening) involves checking the chromosomes of embryos created by in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI)for abnormalities.Embryos with abnormal chromosomes often end in miscarriage, a failed treatment cycle or the child may have a condition like Down’s Syndrome.During PGS, a single cell or a small number of cells is removed from the embryo. The DNA of these cells is then tested to see whether they have any chromosomal abnormalities. Only embryos without chromosomal abnormalities are placed back in the womb.
Who might be recommended to have PGS?
Older women are more likely to have eggs with the wrong number of chromosomes, so traditionally it’s been offered to women over 37. Abnormal chromosomes are thought to be the main reason why older women have difficulties conceiving and are more likely to have a miscarriage or a baby with Down’s Syndrome. Your doctor may also recommend PGS if you have a family history of chromosome problems, if your sperm is at risk of carrying abnormal chromosomes, or if you’ve had several miscarriages or failed IVF attempts without explanation.
Does PGS work?
In the past, some clinics recommended PGS to older women with a history of miscarriage or failed IVF cycles, men whose sperm was at risk of having chromosomal abnormalities or people with a family history of chromosome problems. However, to date there is little evidence showing it improves success rates for these groups. There have been some small clinical trials which have shown that PGS can improve IVF success rates for women under 37 with no history of miscarriage or failed IVF cycles. Until larger trials have been run and we have more evidence, there’s no guarantee that PGS can improve your chances of pregnancy
How safe is it for the embryos?
Because PGS involves removing a cell or number of cells from an embryo this can cause damage to the embryo and prevent it from developing once it has been transferred into the womb. Also research has shown that often the cells in an embryo are not chromosomally identical (called mosaicism). PGS relies on test results from one or a small number of cells being representative of the embryo as a whole. As this is not always the case, it may be possible for an embryo to give an abnormal test result when it is in fact capable of producing a healthy pregnancy.
Are there any other risks?
However, PGS also has some treatment specific risks. This includes the possibility of a misdiagnosis, although modern PGS techniques are very accurate. There’s also the risk that if all the embryos are found to have abnormal chromosomes there won’t be any embryos to put back in the womb. This is especially likely for older women





 WhatsApp us
WhatsApp us